PTFE properties
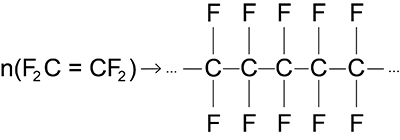
PTFE is obtained through polymerization, by breaking the double bond of tetrafluoroethylene (gaseus) and it has a CF2 structure, from which the name polytetrafluoroethylene.
 PTFE appears in a granular powder at high crystallinity (approx. 95%), with a specific
weight of approximately 2,2 g/cm3 and it reaches a gel form at 327°C (n.b.:PTFE does not
melt!); the molecules have variable dimensions, ranging from 30 to 500 pm based on the
application. Furthermore, particles could be free flow or not free flow.
PTFE appears in a granular powder at high crystallinity (approx. 95%), with a specific
weight of approximately 2,2 g/cm3 and it reaches a gel form at 327°C (n.b.:PTFE does not
melt!); the molecules have variable dimensions, ranging from 30 to 500 pm based on the
application. Furthermore, particles could be free flow or not free flow.
Some of the noteworthy properties of PTFE are:
- very high inertia, due to the presence of fluorine atoms, which, thanks to their
compact structure, shield the carbon atoms of the polymer chain from potential
chemical damages.
PTFE is only undermined by atomic fluorine, trifluoromethyl and
alkali metals at fusion temperatures;
- It is insoluble in every solvent under 300°C;
- It has a wide range of working temperatures (from a minimum of approximately -
268°C to a maximum temperature of 220°C, with peaks of 250°C);
- It is a bad thermal conductor, regardless of the exposure temperature;
- It has extremely low adhesiveness, due to its reduced superficial tension;
- It has the lowest friction coefficient amongst every other existing solid materials;
- It is extremely hydrophobic and completely hydrolysis-proof (it is used as an anti-corrosive barrier and it can hold its characteristics even after years of being in
contact with fluids);
- It is chemically pure and inert;
- It is microbiologically resistant, it doesn’t let bacteria and fungi proliferate;
- It is electrically insulating;
- It is non-flammable;
- It holds up very well against atmospheric and environmental ageing, even when it faces high temperatures or when in contact with solvents, oils, oxidizing agents and UV rays.
- It has good mechanical properties, especially when employed in presence of vibration or flexions.
